Tech #1364: A blood test for detection of congenital heart block in autoimmune-related maternal conditions
Fetal heart block is a life-threatening congenital arrhythmia linked to maternal autoimmune disease, with limited options for prevention despite the large population at risk, including those with conditions such as lupus. Dr. Robert Hamilton has discovered a novel early-pregnancy biomarker and detection method that identifies fetuses at risk before irreversible heart damage occurs, enabling the first true window for preventative intervention.
IP&C is seeking a partnership with either a diagnostic company to complete the development and commercialize the test or with a pharmaceutical company to license the test as a companion diagnostic.
Technology Reference Number
#1364
Inventors
IP&C Contact
Patents
Category
Diagnostic
Keywords
Companion Diagnostic, Maternal, Fetal, Congenital Heart Block
Background
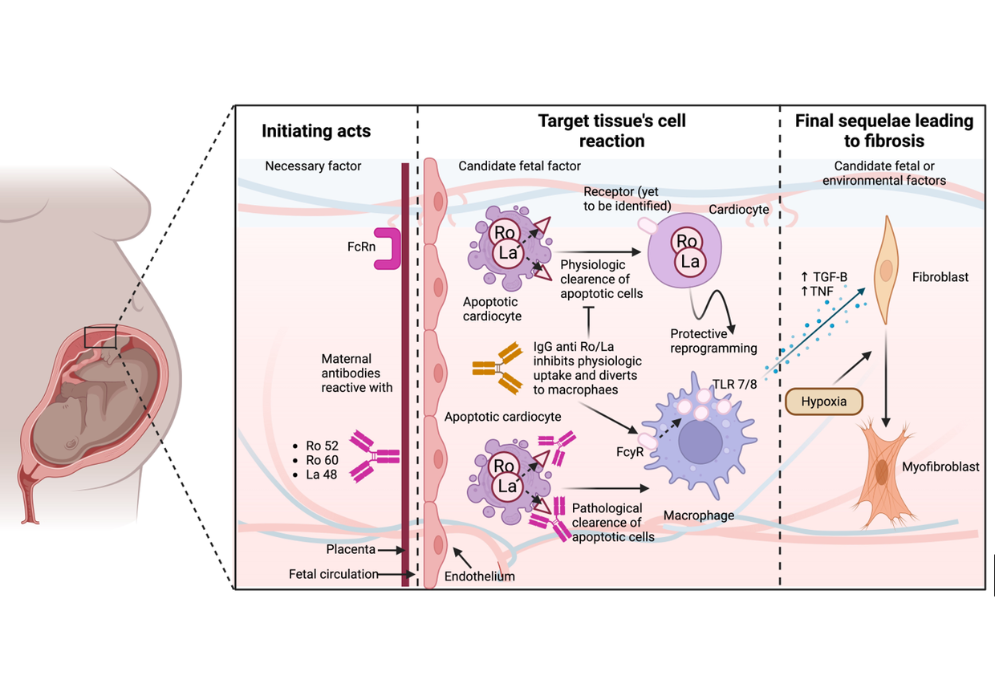
Fetal heart block is a congenital arrhythmia caused by impaired conduction of electrical signals from atria to ventricles, leading to a slowed fetal heartbeat, life-long therapy, or potentially death. Autoimmune congenital heart block is a rare disease but affecting a non-rare population (autoimmune disease) (Lupus affects about 1.5 million Americans and at least 5 million people worldwide, with a higher prevalence in women of childbearing age.) The population is large, the disease is rare, and prevention is currently weak.
Invention Description
SickKids Hospital cardiologist, Dr. Robert Hamilton has identified a novel maternal autoantibody targeting the fetal AT1A1 cardiac protein as a potential biomarker for the early and accurate detection of fetal autoimmune congenital heart block in pregnant women who had not previously had an affected pregnancy.
Commercial Applications
ATP1A1 Changes the Entire Landscape:
- Replaces or reduces fetal echo surveillance by 98–99%.
- Identifies true at-risk pregnancies before disease onset, enabling targeted Plaquenil.
- Predicts CHB well in advance, providing a first-in-class companion biomarker for new therapies.
- Brings number-needed-to-treat/diagnose down to ~1:1.
This advancement might improve the maternal-fetal management of at-risk pregnancies through enhanced surveillance and timely and informed therapeutic interventions.
Current screening fails both sensitivity and specificity:
Anti-Ro Testing
- Misses ~20% of CHB cases.
- Only 1/50–1/100 anti-Ro positive pregnancies develop CHB → very non-specific.
Fetal Echocardiography
- Weekly scans from 16–24 weeks: stressful, expensive, unsustainable.
- Detects disease too late to prevent injury.
- Lifetime cost of pacing a child is extremely high.
Development Stage
In collaboration with a contract research organization, Dr. Hamilton is developing a robust clinical grade assay. Once developed, blood samples from pregnant women across several sites in Europe, Canada and the USA will be assayed in order to test the robustness and validate the accuracy of the assay to detect heart block from diverse ethnicities and genetic backgrounds.