Tech #1386: Innovating Positive Allosteric Modulators (PAM) of potassium channels for drug-resistant epilepsy
Epilepsy affects millions worldwide, yet current antiepileptic drugs are often ineffective or poorly tolerated, leaving many patients without adequate seizure control and facing serious health risks. This technology introduces novel small-molecule positive allosteric modulators of a key voltage-gated potassium channel, offering a targeted approach to restore neuronal stability with greater precision and fewer side effects than existing ion-channel therapies.
IP&C is seeking partnerships to complete preclinical development of the lead compounds and fund medicinal chemistry optimizations before entering IND-enabling studies.
Technology Reference Number
#1386
Inventors
IP&C Contact
Patents
To be filed following optimizations and validation of derivative compounds.
Category
Therapeutic
Keywords
Potassium channel modulator, Drug‑resistant epilepsy, Genetic epilepsy, Neuropsychiatric disorders
Background
Epilepsy affects approximately 65 million people worldwide, with the highest incidence occurring in infancy and early childhood. Despite the availability of numerous antiepileptic drugs (AEDs), treatment outcomes remain poor for a substantial proportion of patients: up to 80% experience significant adverse effects, and approximately 35% develop drug‑resistant epilepsy. These patients are often maintained on complex multidrug regimens, which can exacerbate metabolic, cardiovascular, and neuropsychiatric side effects without adequately controlling seizures. Beyond seizures themselves, epilepsy is associated with increased risk of premature mortality, sudden unexpected death in epilepsy (SUDEP), injury, and high rates of comorbid psychiatric disorders such as depression and anxiety. There is therefore a critical need for novel, targeted therapies that address neuronal hyperexcitability with improved efficacy and safety, particularly for patients with treatment‑resistant or genetically driven epilepsies.
Invention Description
This technology centers on the development of novel, small‑molecule positive allosteric modulators (PAMs) of the target voltage‑gated potassium channel. This channel plays a fundamental role in shaping action potentials, regulating neuronal firing, and maintaining excitatory/inhibitory (E/I) balance in the brain. Loss‑of‑function or hypoexpression of is directly linked to juvenile epilepsy.
The class of compounds discovered, act as positive allosteric modulators that directly interact with a previously unexploited druggable cavity discovered by in silico docking experiments. Structural and electrophysiological studies show that these molecules bind within this cavity to enhance channel opening, lower the activation threshold, and shift the voltage dependence of activation toward more hyperpolarized potentials. By selectively enhancing potassium channel function rather than broadly blocking ion channels, this approach aims to normalize neuronal excitability while avoiding the liabilities seen with sodium, calcium, or non‑selective potassium channel modulators.
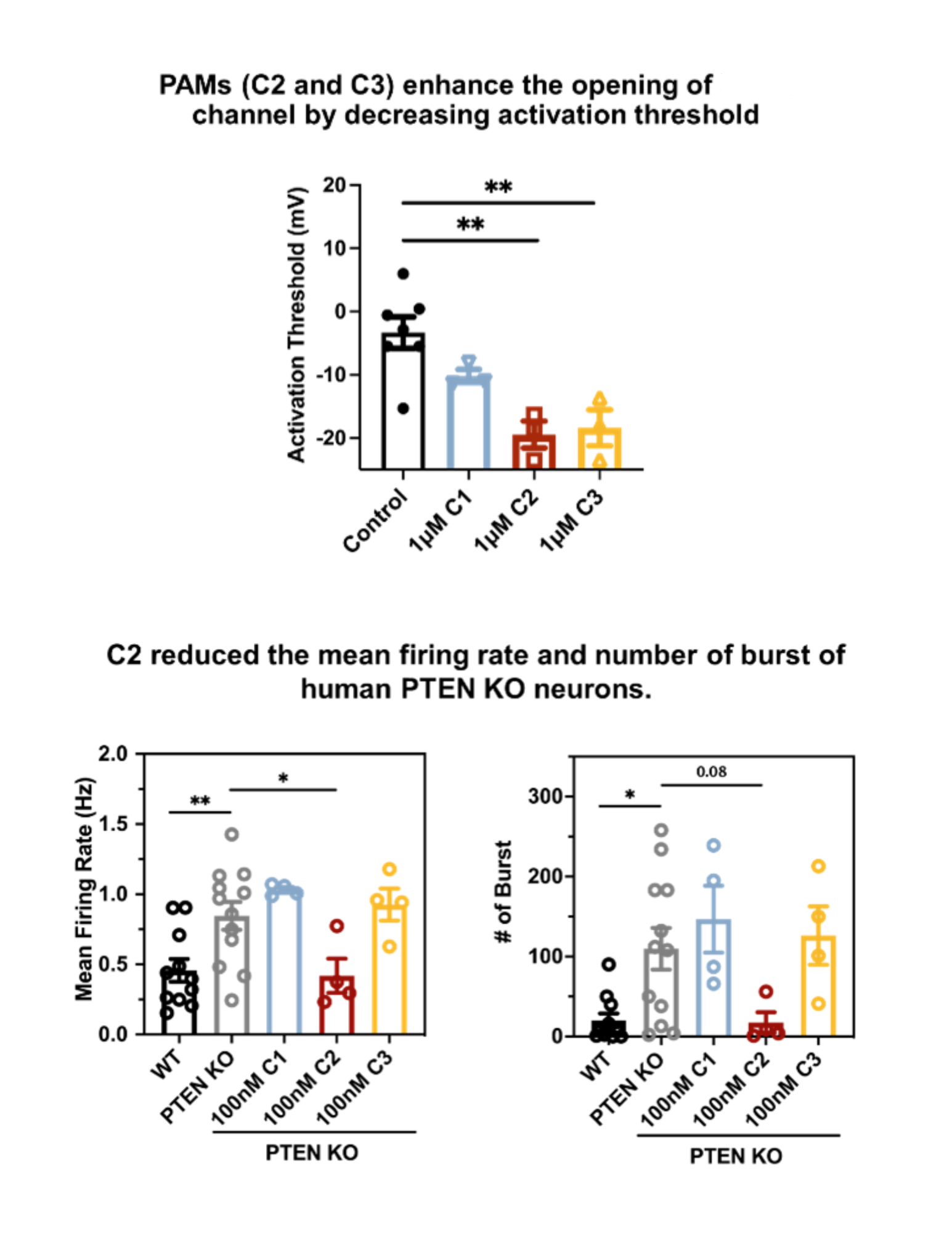
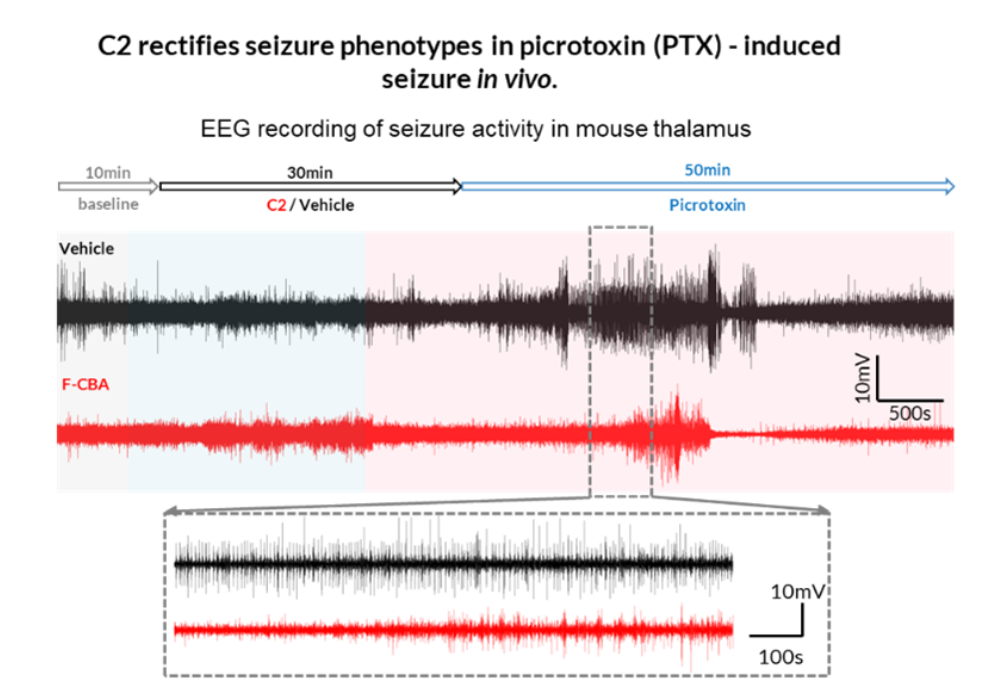
Figure 1.
Compound C2 and its analogs potentiate channel activity to suppose epileptic phenotypes in mutant cells and PTEN KO neurons.
Development Stage
In vitro electrophysiology demonstrates that compounds C2 and C3 potently enhance channel currents, reduce neuronal activation thresholds, and suppress pathological firing patterns (Figure 1). In patient‑relevant, drug‑resistant epilepsy models, these compounds rescue hyperexcitability in PTEN knockout human iPSC‑derived neurons, as shown by multi‑electrode array recordings with marked reductions in firing rate and burst activity.
In vivo efficacy has been demonstrated across multiple seizure models. C2 significantly delays seizure onset and shortens seizure duration in picrotoxin‑induced seizure models, accompanied by reduced epileptiform EEG activity. In genetic disease contexts, repeated dosing of C2 and C3 is well tolerated and effective in Fmr1 knockout mice, an autism spectrum disorder model that exhibits audiogenic seizures, supporting both antiseizure and broader neurobehavioral relevance. Across studies to date, the lead compounds show no overt toxicity following acute or chronic administration, with evidence of systemic exposure and brain penetration.
Ongoing medicinal chemistry efforts are focused on optimizing analogs to further improve blood–brain barrier permeability and oral bioavailability.
Commercial Applications
This technology supports a multi‑indication clinical and commercial strategy. The initial focus is precision therapy for genetic epilepsies, including patients with PTEN, and FMR1 mutations that are underserved populations with limited or no approved targeted treatments. Beyond rare genetic indications, the PAMs represent a compelling, first‑in‑class approach for the much larger treatment‑resistant epilepsy population, which accounts for approximately one‑third of all epilepsy patients globally.
Preclinical evidence also supports expansion into neuropsychiatric disorders, particularly depression and anxiety, which are highly prevalent comorbidities in epilepsy and remain inadequately treated by existing AEDs. Notably, no other groups are currently developing positive allosteric modulators against the target channel, creating a differentiated competitive position within a validated ion‑channel therapeutic paradigm. Together, these features position the PAMs with commercial applications spanning rare, refractory, and common CNS disorders.